Total Control
Know exactly which prints exist, who created them, and whether they were approved.
Traceable, compliant, secure
In many regulated industries — from pharmaceutical manufacturing to biotech and laboratories — digital data is well controlled. Validated systems such as LIMS, QMS, or ERP safeguard registration and traceability.
Yet the moment a document is printed, the process often falls outside that controlled chain. QA teams still manually approve, number, and record batch records to maintain traceability — a time-consuming and error-prone process. A wrong or missing print, a duplicate, or an unverified copy can lead to audit findings or delays in batch release.
TR-Print closes this gap — securing the last uncontrolled step between digital data integrity and paper-based process control.
When printing is not controlled, your audit trail is incomplete — even if your digital systems are validated.


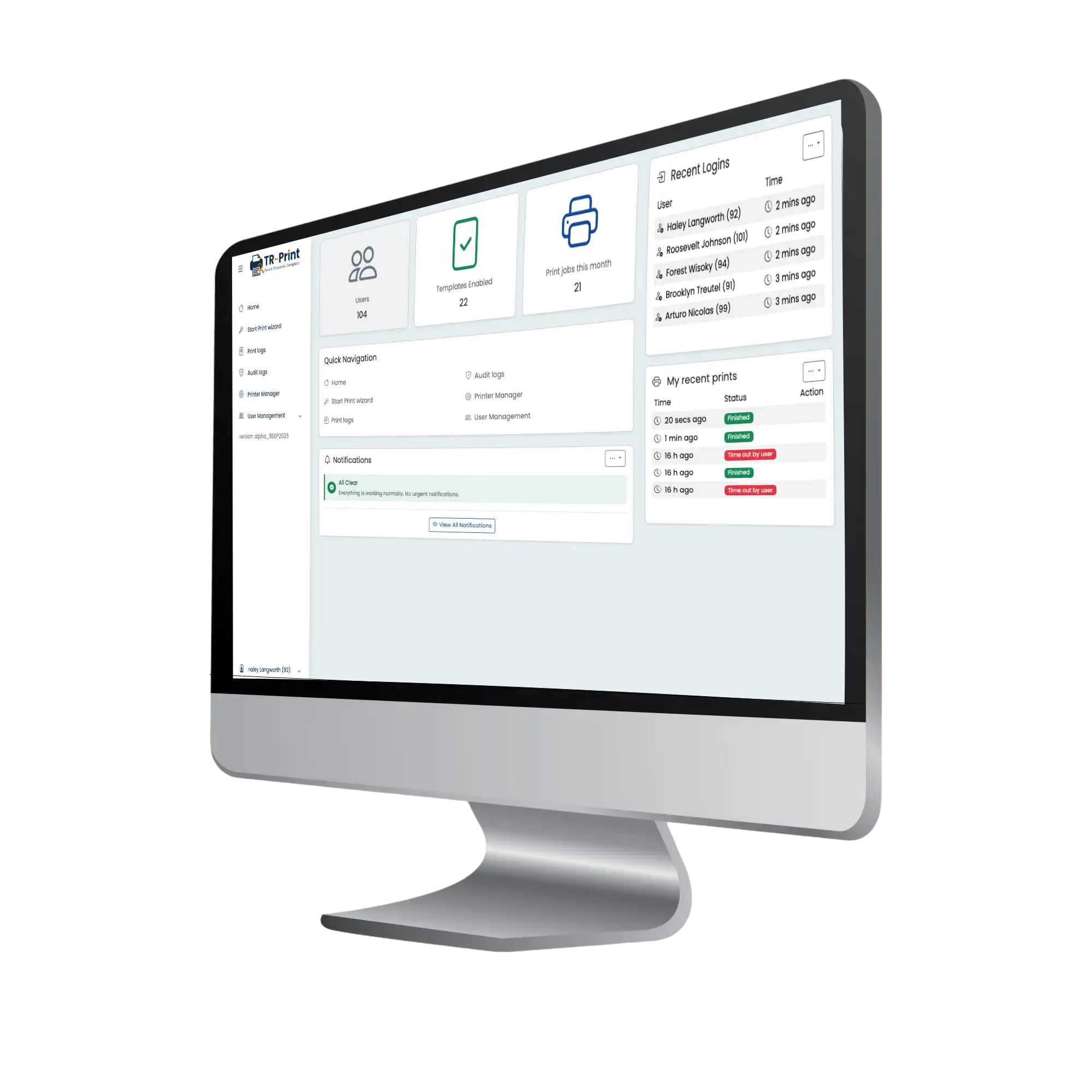
TR-Print is not a generic print tool, but a specialized application that brings the printing of test and batch records under full quality and compliance control — available as SaaS, on-premise, or in a hybrid configuration.
Within TR-Print, organizations upload approved document templates — such as test records, batch forms, or certificates of analysis. From these templates, controlled prints are executed, automatically assigned with:
After printing, the user confirms whether the printout was successful and meets physical quality requirements. Unreadable or defective prints can be marked as failed, and TR-Print records this automatically. The result: a complete and reliable audit trail showing exactly which records are in circulation, which failed, and which are pending.
TR-Print is available as SaaS, on-premise, or in a hybrid configuration — whichever fits your organisation's data and IT requirements. In every setup, a small TR-Print Node is placed on site behind the printer to create a secure print connection. No complex IT setup required.
Upload validated versions of your test or batch forms into TR-Print. Only approved templates can be used for controlled printing.
Each print job receives a unique ID and sequential numbering. All details — user, time, printer, and result — are logged automatically.
The user confirms that the print was produced correctly and is readable. Any failed or rejected print is flagged instantly, keeping your reconciliation up-to-date.
All print data is securely archived. Every document, user, and deviation remains traceable — anytime.
One source. One ID. One auditable truth.
Ready to remove printing as a compliance risk in your quality process?
Starting from €700/month · SaaS or on-premise · No complex IT setup
Each printout is uniquely identifiable — eliminating duplicates, uncontrolled copies, and undocumented reprints
Users verify that each print is successful and of acceptable quality. Failed prints are automatically logged.
Only approved templates can be printed — ensuring consistency and version control.
All activities are logged automatically and instantly available for audits.
Automatic reports per batch, user, or time period — always inspection-ready.
Available as SaaS, on-premise, or hybrid — manage templates, printers, and user rights centrally in every configuration.

In GMP environments, printed test records must always be fully accounted for. Auditors expect clear proof that all issued records are either returned, archived, or properly explained. TR-Print automatically performs end-of-process reconciliation, giving full visibility over the lifecycle of every printed test record.
With TR-Print you can instantly see:
Because every record receives a unique, non-reusable ID, missing records can always be traced back to the originating process or user. This level of reconciliation and traceability is highly valued during GMP and data-integrity audits.
TR-Print is designed for organizations operating under GMP, ISO, or FDA regulations where printed documentation remains part of the quality process:
QA teams need certainty — not assumptions. TR-Print eliminates manual print controls, ensures full traceability, and standardizes documentation integrity across sites. Less manual work, fewer audit findings, faster review cycles.

Printing used to be the last uncontrolled step in regulated environments — TR-Print closes that gap. It brings accountability, traceability, and reconciliation to every printed record, without changing your workflow. Simple to deploy, easy to use, and built for GMP compliance. Confidence in every print — from first copy to final reconciliation.
TR-Print is built for regulated environments where paper is still part of the validated process.
Know exactly which prints exist, who created them, and whether they were approved.
Available as SaaS, on-premise, or hybrid — minimal IT effort in every configuration. Just one TR-Print Node per printer, no complex rollout required.
Real-time dashboards and reports simplify audit preparation and batch release.
Every document is unique, traceable, verified, and confirmed — removing doubt during inspections.
Schedule a live walkthrough with our team. We'll show you how TR-Print fits into your current quality process — no lengthy RFP, no IT project required.
Traceable printing means every printed document — such as a test record, batch record, or label — is uniquely identified, numbered, and logged. In GMP environments, this is essential for data integrity and compliance with regulations like Annex 11 and 21 CFR Part 11. TR-Print ensures every printout can be traced back to the user, printer, and approved template — eliminating uncontrolled copies.
TR-Print automates secure printing processes, applies unique document IDs, and guarantees reconciliation of printed records. This makes every print traceable and always audit-ready for GMP, Annex 11, and FDA 21 CFR Part 11 inspections.
TR-Print enforces data integrity by making the printing process itself controlled and auditable. Every print action is logged with who printed what, when, and why. Failed prints, duplicates or reprints are automatically tracked and included in reconciliation reports — supporting ALCOA+ principles of data integrity (Attributable, Legible, Contemporaneous, Original, Accurate).
Yes — TR-Print was designed specifically for GMP and pharma traceable printing. It ensures that all printed batch records, labels, and analytical forms are produced from approved templates and remain fully traceable throughout the lifecycle. This helps QA departments demonstrate compliance during inspections and audits.
By default, TR-Print is a SaaS platform cloud-based with a small local TR-Print Node behind each printer. For organizations with strict data policies, on-premise and hybrid configurations are available. All data traffic is encrypted, and user authentication integrates with your existing security model.
TR-Print aligns with key global standards for regulated industries:
This makes TR-Print suitable for pharmaceutical, biotech, medical device and laboratory environments that require traceable GMP printing.
Chroomstraat 40, 2718RR
Zoetermeer, The Netherlands
Monday - Friday
9:00 - 17:00 (CET/CEST)
This form is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service are in effect.